Home

The Possibilities of Renewable Propane
Posted on March 13, 2024
With Low Carbon Fuel Standards, Clean Heat Standards and similar laws under…
Propane Can Do That
Posted on March 12, 2024
Take Action and Take Control of Your Future at Industry Summit VI and Visions ‘24
Posted on March 11, 2024

RINs and Repeat
Posted on March 12, 2024
Volatility in the RINs market directly affects liquid heating fuel companie…
ISO NE – Not Ready for Prime Time?
Posted on March 11, 2024
U.S. DOE Releases Home Energy Rebate Guidance
Posted on September 14, 2023
Canada Passes Clean Fuel Regulation
Posted on September 13, 2023

Optimizing Operations
Posted on February 12, 2024
Best practices for terminal and fuel transport operators
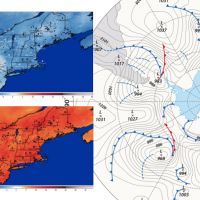
National Weather Forecasting Takes NEFI by Storm
Posted on December 8, 2023
A Chance Hydrogen Discovery
Posted on December 7, 2023
Winter 2023-2024 First Impressions
Posted on October 11, 2023

NORA Net-0 Home Update
Posted on October 6, 2023
The need to lower carbon emissions has driven many industries to find alter…
Building Your Information Network with Propane Tank Monitors
Posted on August 11, 2023
A Recipe for Delivery Efficiency
Posted on August 11, 2023
Don’t Ignore the Storage Capacity You Already Own
Posted on July 18, 2023

Carlin Begins Production on UL Listed B100 Burners
Posted on December 7, 2023
Carlin Combustion Technology has announced the launch of their B100 burner
NORA Looks at Indoor Air Quality
Posted on December 7, 2023
Why Market Renewable Propane for Your Business?
Posted on October 6, 2023
Are Carbon Handprints a Better Metric?
Posted on October 6, 2023

The Four Key Metrics of Your Hedging Strategy
Posted on March 12, 2024
It’s a good time to look back before looking forward!
Market Forecast: What to Watch
Posted on February 12, 2024
Wrangling Cash Market Differentials and Crack Spreads
Posted on December 7, 2023
Is It Déjà Vu All Over Again?
Posted on October 10, 2023
Fill Your Idea Parking Lot for 2024
Posted on February 12, 2024
Known Knowns and Unknown Unknowns
Posted on December 7, 2023
Expanding the Definition of Equipment Sales
Posted on October 10, 2023

Innovative Financing Solutions for Energy Businesses
Posted on March 12, 2024
Growth capital doesn’t need to come from a traditional bank loan
Service, Support and Collaboration Separate Payment Partners From a Payment Processor
Posted on March 12, 2024
Heating Things Up: How Influencer Marketing Will Ignite the Oil Heat Industry
Posted on March 12, 2024
Capital Allocation Strategies for Sustained Growth in an Energy Business
Posted on February 12, 2024
Join Our Email List For Updates!
Enter your email to receive important news and article updates.
Industry Prices